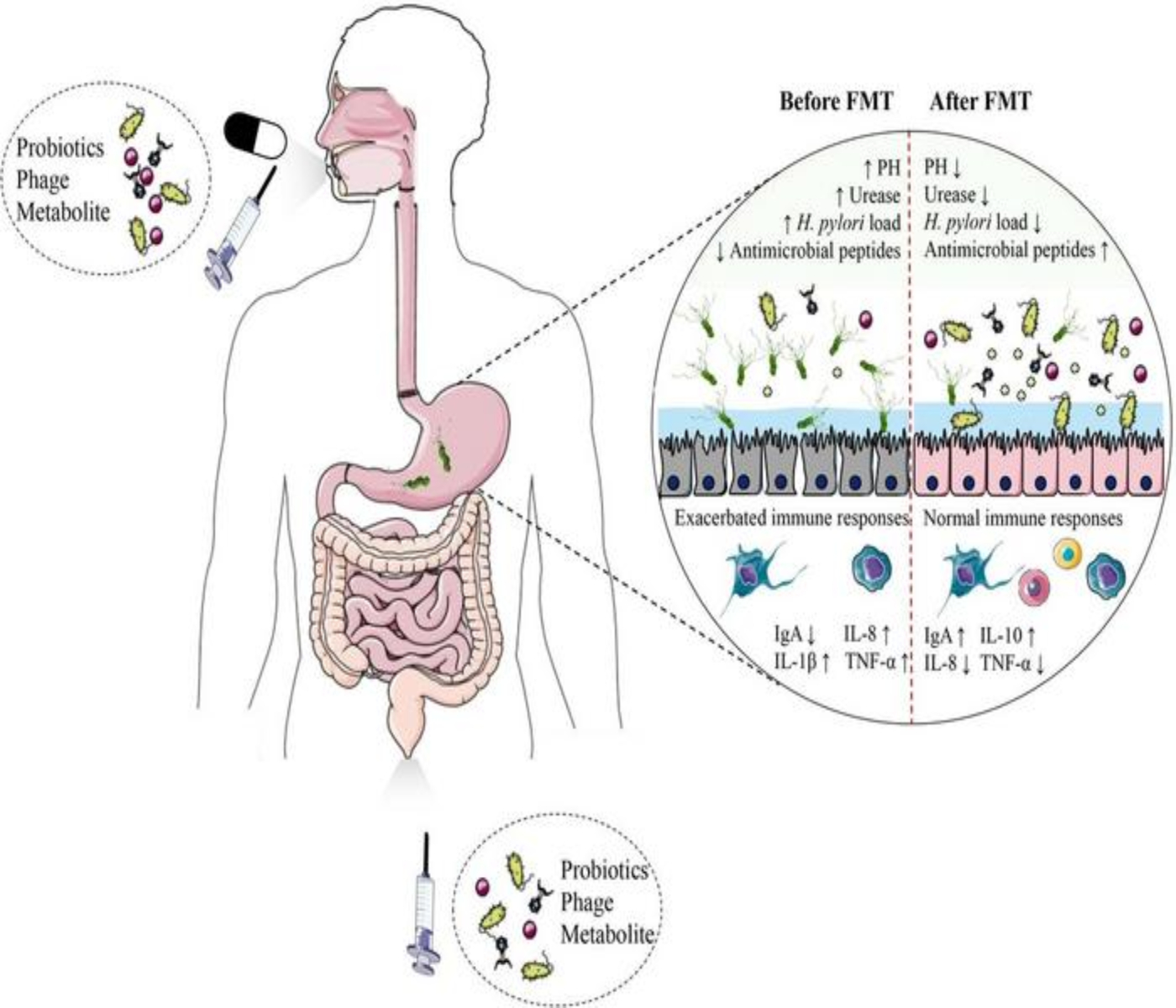
The very supplements farmers use to keep their livestock healthy might be silently contributing to a global health emergency. Startling new research from China reveals that many probiotic products for animals, intended as a safe alternative to antibiotics, are not only falling short on quality but are also acting as unwitting carriers of antibiotic resistance. This groundbreaking study, published in Engineering, casts a shadow over the widespread use of these supplements, exposing a troubling connection to the rise of drug-resistant “superbugs.”
For years, beneficial bacteria like those found in probiotics have been touted as a way to boost animal health without relying on traditional antibiotics, a practice that can foster drug resistance. This investigation, however, delivers a harsh dose of reality: many of these products are mislabeled, contain too few beneficial bacteria, and, most critically, harbor dangerous genes that enable bacteria to resist life-saving antibiotics. These resistance genes can then spread to other bacteria, including those that cause serious illnesses in humans. What we give our farm animals can profoundly affect our own health and the effectiveness of our medicines.
Unmasking the Invisible Danger: What the Study Uncovered
Researchers in China embarked on a comprehensive mission, collecting 95 different probiotic products intended for livestock. These products, gathered from across 22 Chinese provinces between 2021 and 2023, offered a wide view of what was truly on the market. Their investigation wasn’t just about checking labels; it was a deep dive into the actual contents, bacterial counts, and potential hidden dangers within these supplements.
The first step involved isolating and identifying the bacteria. Scientists grew samples from each product on special plates to pinpoint species like Bacillus, Enterococcus, and Lactobacillus. They used advanced genetic fingerprinting (a process that analyzes specific DNA sequences, like the 16S ribosomal RNA gene) to confirm the identity of each bacterial type. Beyond the “good” bacteria, they also meticulously searched for any unwelcome guests—bacteria that could cause disease if given the chance.
Once identified, these bacterial strains faced a crucial test: exposure to common antibiotics. This “drug resistance test,” also called antimicrobial susceptibility testing, showed which bacteria could shrug off the effects of vital medications. The antibiotics tested included those critical for treating infections in both animals and humans, such as tetracycline and erythromycin.
To truly understand the threat, researchers then performed “whole genome sequencing.” This process reads the entire genetic blueprint of each bacterial strain, allowing them to pinpoint specific “antibiotic-resistance genes” (ARGs), which are segments of DNA that give bacteria the ability to defeat antibiotics, and “virulence genes,” genes that make bacteria harmful. They also looked for “mobile genetic elements” (MGEs), which are like tiny, self-replicating pieces of DNA that can easily jump from one bacterium to another, carrying resistance genes along for the ride. This mobility is key to how antibiotic resistance spreads.
The team also conducted “gene transfer experiments” in the lab (known as conjugative assays) to see if these resistance genes could indeed jump between bacteria. They specifically focused on genes like optrA, which grants resistance to a class of crucial, last-resort human antibiotics called oxazolidinones. To confirm these lab findings, they then set up an experiment with 105 newly hatched broiler chickens. Some chickens received an Enterococcus strain known to carry the optrA resistance gene, others a similar strain without it, and a control group received none. For 35 days, the researchers monitored the chickens’ gut bacteria and the presence of resistance genes. This real-world experiment, performed “in vivo” (in a living organism), provided powerful evidence of how these resistance genes could spread within a living system.
Troubling Discoveries: Quality, Contamination, and Spreading Resistance
The study’s findings are deeply unsettling, highlighting significant problems in the current probiotic market for livestock in China.
Widespread Quality Issues: Many probiotic products simply didn’t live up to their claims. For instance, if a product claimed to contain Lactobacillus, there was an alarming 89% chance it either didn’t contain it or didn’t have enough to be effective. While Bacillus and Enterococcus products fared better, there were still notable discrepancies between label promises and actual content.
Even when the correct bacteria were present, their numbers were often too low. Most products aim for a minimum of 100 million viable bacteria per gram (a measure often expressed as 1.0 × 10⁸ CFU per gram, where CFU stands for Colony-Forming Units) to be effective, yet only about 73% of Bacillus products met this, and Enterococcus and Lactobacillus products showed even lower success rates, at roughly 62% and 38%, respectively. This indicates many supplements likely aren’t delivering the intended health benefits.
Perhaps the most alarming discovery was that approximately one-third of the probiotic products were contaminated with “opportunistic pathogens.” These are bacteria that don’t usually cause illness in healthy individuals but can become dangerous under certain conditions, such as a weakened immune system. The researchers identified several such contaminants, including Bacillus cereus, Enterobacter cloacae, Klebsiella pneumoniae, and Salmonella enteritidis. Many of these contaminants also carried genes that make them more harmful, such as toxins that can cause food poisoning.
The Alarming Spread of Antibiotic Resistance: The study singled out Enterococcus bacteria as a major concern. While other beneficial bacteria showed some natural resistance to antibiotics, Enterococcus strains displayed a far more complex and worrying pattern of resistance to multiple drugs. This indicated they had acquired these resistance genes from their environment, rather than having them naturally.
A particularly dangerous gene, optrA, was identified in one Enterococcus faecium isolate. This gene confers resistance to a class of “last-resort” antibiotics used to treat severe human infections. Crucially, this optrA gene was found on “novel transferable plasmids”—small, circular pieces of DNA that can easily jump between different bacteria. These plasmids act as potent vehicles for gene transfer.
The chicken experiment provided compelling proof that this optrA gene is indeed mobile. It jumped from the probiotic Enterococcus to other bacteria commonly found in the chicken’s gut, including E. cecorum, E. gallinarum, and L. crispatus. This transfer significantly increases the overall pool of antibiotic-resistance genes within the chickens. Analysis of the genetic material in the chickens’ guts (metagenomic analysis) further confirmed that the optrA gene could be transferred via a specific mobile genetic element called transposon IS1216E to these common gut bacteria.
Even more concerning, some of the Enterococcus strains (specifically E. faecium ST262 and ST396, and E. faecalis ST16) isolated from these animal probiotic products were genetically very similar to Enterococcus strains known to cause serious infections in humans, such as wound and bloodstream infections. This close genetic link raises the disturbing possibility of these resistant strains directly impacting human health.
The Far-Reaching Consequences: From Farms to Our Plates
The implications of these findings are substantial. When livestock are given probiotic supplements containing these rogue, antibiotic-resistant bacteria, those bacteria can set up residence in the animals’ digestive systems. From there, the resistance genes can readily transfer to other bacteria within the animal, including those that are normally harmless.
This creates a hidden reservoir of antibiotic resistance within the livestock population. These resistant bacteria, or their mobile resistance genes, can then spread through several avenues:
- Through the food we eat: Consuming meat from these animals poses a risk of ingesting these resistant bacteria. While proper cooking typically kills bacteria, improper handling or undercooked meat could lead to exposure.
- Environmental contamination: Animal waste containing resistant bacteria can pollute soil and water, further spreading antibiotic resistance into our ecosystem.
- Direct contact: Farmers and agricultural workers who interact with livestock could also be exposed.
The true concern goes beyond direct infection. It contributes to the broader problem of antibiotic resistance, a global health crisis that threatens our ability to treat common infections. Each time an antibiotic-resistance gene spreads, it weakens the effectiveness of existing drugs, pushing us closer to a future where even minor infections could become deadly.
A Critical Need for Oversight and Safety
This study serves as a powerful reminder that “natural” doesn’t always mean safe or effective. The widespread use of low-quality Enterococcus strains, especially those carrying antibiotic-resistance genes, in livestock probiotics could disrupt the natural balance of gut bacteria in animals and dangerously accelerate the spread of these resistance genes.
The message is clear: the industry must rigorously evaluate the safety and quality of probiotic products, particularly those containing Enterococcus strains. This means more than just checking labels; it requires ensuring the actual bacterial content, viable cell counts, and, critically, the absence of harmful contaminants and transferable antibiotic-resistance genes. For consumers, it underscores the importance of safe food handling practices and a deeper understanding of how animal health, environmental health, and human well-being are inextricably linked. This research, firmly rooted in the principles of “One Health” and “food safety,” provides crucial evidence for a more regulated and responsible approach to probiotic use in livestock.
Paper Summary
Methodology
The study analyzed 95 commercial livestock probiotic products from 22 Chinese provinces (2021-2023). Researchers identified bacterial strains and opportunistic pathogens using culture and genetic sequencing (16S rRNA, whole genome sequencing). They tested for antibiotic resistance, identified resistance and virulence genes, and analyzed mobile genetic elements. Gene transfer was assessed in lab experiments (conjugative assays) and a 105-chicken model over 35 days, specifically observing optrA gene transfer in the gut.
Results
Significant quality control issues were found, including low Lactobacillus labeling compliance (11%) and product contamination (33.3%) with opportunistic pathogens carrying harmful genes. Enterococcus strains showed acquired multidrug resistance, with the optrA gene found on transferable plasmids. The chicken study confirmed optrA transfer via transposon IS1216E to common gut bacteria (E. cecorum, E. gallinarum, L. crispatus), disrupting normal microbiota. Some Enterococcus strains were genetically similar to human infectious clones.
Limitations
This “Journal Pre-proof” is subject to further editing. Findings are specific to Chinese probiotic products and may not be broadly generalizable. While ARG transfer was shown in chickens, the study notes that further large-scale, long-term research is needed for full ecological impact understanding.
Funding and Disclosures
The paper is open-access under a CC BY-NC-ND license. Authors are affiliated with Jiangsu Key Laboratory for Food Quality and Safety & State Key Laboratory Cultivation Base of Ministry of Science and Technology, Institute of Food Safety and Nutrition, Jiangsu Academy of Agricultural Sciences, and Jiangsu Co-Innovation Center for Prevention and Control of Important Animal Infectious Diseases and Zoonoses, College of Veterinary Medicine, Yangzhou University, China. The animal study was approved by the Jiangsu Academy of Agricultural Sciences (IACUC-AE-2023-08-010).
Publication Information
Title: Livestock Probiotics in China: Quality Analysis and Enterococcus-Associated Antibiotic Resistance Dissemination Risks Authors: Xing Ji, Jiayun Wang, Jun Li, Lili Zhang, Ruicheng Wei, Ran Wang, Tao He Journal: Engineering PII: S2095-8099(25)00196-1 DOI: https://doi.org/10.1016/j.eng.2025.03.032 Received Date: 16 October 2024 Revised Date: 4 February 2025 Accepted Date: 6 March 2025 Published By: Elsevier LTD on behalf of Chinese Academy of Engineering and Higher Education Press Limited Company